 Re: Physical meaning of positive and negative net charge ( No.1 ) Re: Physical meaning of positive and negative net charge ( No.1 ) |
- Date: 2024/03/30 11:08
- Name: Vipin Kumar
- Dear Y.-T. Lee,
Could you please help me in this regard?
Thank you very much.
Regards,
Vipin
|
 Re: Physical meaning of positive and negative net charge ( No.2 ) Re: Physical meaning of positive and negative net charge ( No.2 ) |
- Date: 2024/03/31 04:31
- Name: Yung-Ting Lee
- Dear Vipin,
Q1: I think that your explanation is correct. Or, you can test a CO2 molecule. The net charge result will be the same as your explanation.
Q2: A simple case can be
(1) Calculate net charges of an isolated CO molecule and an isolated WS2 surface.
(2) Calculate net charges of a CO molecule adsorbed on WS2 surface.
Check the net charge difference of atoms between (CO/WS2 surface) and (CO adsorbed on WS2 surface).
One may know the net charge difference easily.
Best regards,
Yung-Ting Lee
|
 Re: Physical meaning of positive and negative net charge ( No.3 ) Re: Physical meaning of positive and negative net charge ( No.3 ) |
- Date: 2024/03/31 15:32
- Name: Vipin Kumar
- Dear Lee,
Thank you very much for your prompt response. I request you to clear one more doubt:
I have calculated the net charge on individual atoms in CO adsorbed WS2 using net charge scheme available in the OpenMX viewer. I get the following
charge on W=0.375 and S=-0.198 Then, the net charge on WS2 will be equal to (net charge on W-atom+2xnet charge on the S-atom)=0.375+2x-0.198= -0.021. That means the net charge on WS2 will be -0.021e.
Similarly, for CO molecule, it will be (net charge on C-atom+net charge on O-atom)=0.320 + (-0.365)=-0.045e, then the net charge on CO molecule is -0.045e.
Is it the right way to calculate the net charge on WS2 and CO molecule? Any help in this regards will be appreciated.
Thank you very much.
Regards,
Vipin
|
 Re: Physical meaning of positive and negative net charge ( No.4 ) Re: Physical meaning of positive and negative net charge ( No.4 ) |
- Date: 2024/04/01 05:27
- Name: Yung-Ting Lee
- Dear Vipin,
(1) One may notice that the net charge of S atom bonding with CO is different from that of S atom bonding without CO.
After CO adsorption, S atoms' net charge should be different.
One can not treat S=-0.198 e for all S atoms when CO adsorption on WS2 surface structure.
(2) For CO adsorption on a WS2 surface, CO is connected to S atom(s) (the surface layer / the top layer), not W atom(s) in the middle layer and the other S atom(s) in the bottom layer.
For the discussion on charge redistribution during CO adsorption on the WS2 surface, consider focusing on atoms that form bonds between CO and S atom(s).
(3) You can check how other researchers usually define/calculate the net charge for an atom or a molecule on a surface from their published Journal papers.
Also, textbooks often give some examples.
(4) For your reference,
Viet Q Bui et. al. discussed about "CO adsorption on a WS2 monolayer" in 2015 in J. Phys.: Condens. Matter 27, 305005.
Table 3 is "Charge transfer from the WS2 layer to gas molecules (CO, H2O, NO, and O2)".
Best regards,
Yung-Ting Lee
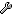 |
 Re: Physical meaning of positive and negative net charge ( No.5 ) Re: Physical meaning of positive and negative net charge ( No.5 ) |
- Date: 2024/04/01 13:16
- Name: Vipin Kumar
- Dear Lee,
Thank you very much for your help.
With regards,
Vipin
|
|
 This thread is locked.Only browsing is available.
This thread is locked.Only browsing is available. This thread is locked.Only browsing is available.
This thread is locked.Only browsing is available.